What is Regulatory Information Management System (RIMS)?

Regulatory Information Management Systems Software (RIMS) are centralized platforms designed to support and streamline Regulatory Affairs teams and operations. RIMS helps in the creation, organization, submission, and archival of regulatory information in regulated formats.
The process is designed to manage and store information in a traceable fashion to the specified local markets and ICH Guidelines pertinent to the registration and approval of a product. It enables end-to-end tracking, managing regulatory activities and objects, and archiving respective product dossiers or data for future use.
RIMS’ existence dates back to the medicinal products regulations’ inception. However, the standardization of the current processes that harmonize regulatory submission in different geographic areas was formulated by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) in collaboration with the FDA, the EMA, and other ICH members in the year 2000.
A common set of documentation known as the Common Technical Document (CTD) for approval submissions was then created to ease regulatory information complexities.
What Is the Role of RIMS in Regulatory Compliance?
Regulatory compliance is an ongoing process; thus, mismanagement of regulatory information can be detrimental to an organization’s growth and innovation, derailing the product to market access and consequently implicating the organization’s market share.
The traditional approach of multiple spreadsheets, disintegrated systems, and SharePoint, silo working teams and can no longer accommodate the dynamic and evolving global regulatory submission requirements.
Clinical trials and product registration regulations are becoming more complex; successful teams must be harmonized and have clear, transparent workflows and integrated systems to achieve compliance, positive regulatory audits, and successful product development.
An information system that provides data integrity and continuity, plans, manages, tracks, and archives regulatory submissions and Health Authority (HA) correspondences, is the first step in managing the risk associated with evolving health authorities’ laws.
Integration of RIMS with other systems, such as publishing tools, document management systems, quality management systems, etc., is of paramount importance.
Furthermore, regulatory affairs teams must adequately develop processes and best practices for data/document management to optimize RIMS use and reduce potential business risks.
The Core Functions of RIMS
RIM core operations can be divided into three main functions:
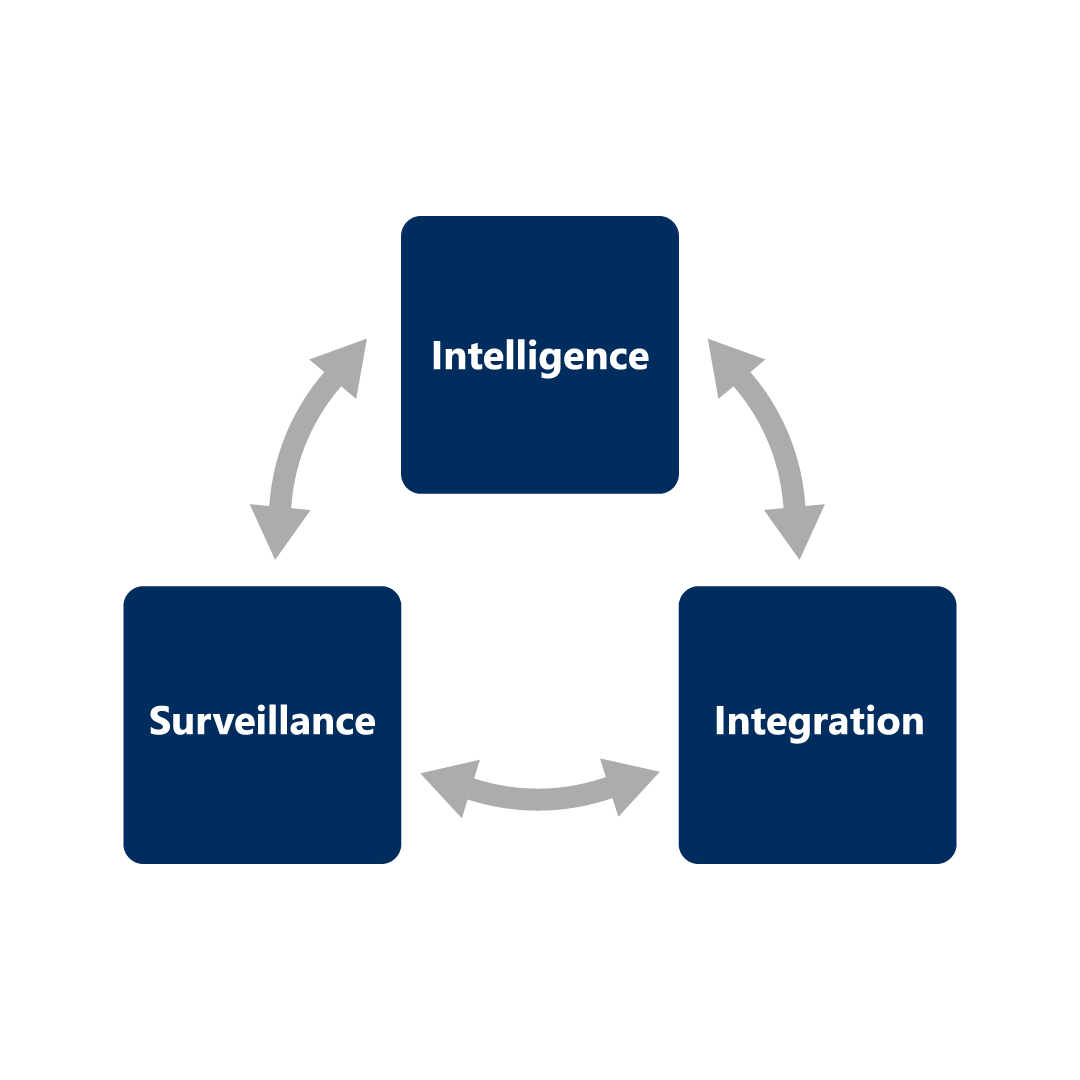
Intelligence
- Facilitates regulatory strategic planning, Authoring, Dossier Creation, Dispatch archiving, tracking of submissions, HA interactions
- Product development analysis and global market access
Integration
- Creation of workflow to facilitate streamlined approach for cross functional teams
- Interface with other business systems to provides transparency and a centralized approach
Surveillance
- Generating and monitoring reports and dashboards
- Safety reports, risk management, and mitigation process monitoring
Why Are Sponsors Required to Use RIMS?
Most organizations are multinational companies, and their data collation, processing, and organization can be tedious. A RIM system can tremendously impact all organizational functions by centralizing information, automating, and streamlining submission activities that can also be easily backtracked.
The below few points are observed for organizational support:
- An efficient RIM system fast-tracks the product-to-market process for Sponsors, thereby providing a competitive edge.
- Standardizes & simplifies lifecycle management of products or documents from clinical trials to postmarket authorization, ensuring regional regulatory requirements and laws are met.
- Information is seamlessly tracked, approved, and archived, reducing silo working within an organization.
- Streamlines global regulatory compliance.
- Provides transparency during the auditing a Sponsor’s multinational regulatory activities and operations.
Outsourcing RIM Functional Deliverables
Some Sponsors need more resources or RA experts who can dedicate their time to follow and meet the requirements for RIMS within the given timelines or ongoing basis. Skilled professionals are needed to understand, manage, and control submission tracking and archival of health authority correspondences and commitments.
Leveraging a CRO with expertise in this space will ensure that a dedicated team is assigned to manage RIMS activities and supplemental support to their RA teams.
Some organizations just beginning to transition into a RIM system may need experts to advise on setting up processes, assist with non-compliance, and create an effective and efficient RIMS process right from the start.
Organizations with comparatively huge portfolios can also look for external expertise within RIMS to continuously improve processes and pioneer themselves within industry trends.
If you have any questions about RIMS, or would like to speak to a regulatory operations expert, please email info@mmsholdings.com or visit our website.
By: Nomakhwezi Mvumvu, Associate Manager of Regulatory Operations, and Vidyasagar Chowdary Vadde, Team Lead, Regulatory Operations











