FDA REMS Programs, Considerations, and Compliance

The Food and Drug Administration (FDA) Amendments Act of 2007 authorized the FDA to require a Risk Evaluation and Mitigation Strategy (REMS) Program. The REMS program is required if it is determined that specific safety measures are needed to ensure that a drug’s benefits outweigh the risks, either during the initial product review or at any point in the post-marketing period. REMS Programs may be required for a single drug or broadly across a general classification of drugs, which share risk-benefit components.
Before 2007, sponsors were issued a Complete Response Letter (CRL), which left potential drugs in deferred regulatory approval indefinitely until additional safety data could be obtained. Pre-approval REMS requirements have provided avenues to obtain additional safety data under REMS requirements. Post-approval REMS are typically requested when new safety information determines the necessity to ascertain if the drug benefits outweigh the safety risks.
REMS Program Considerations
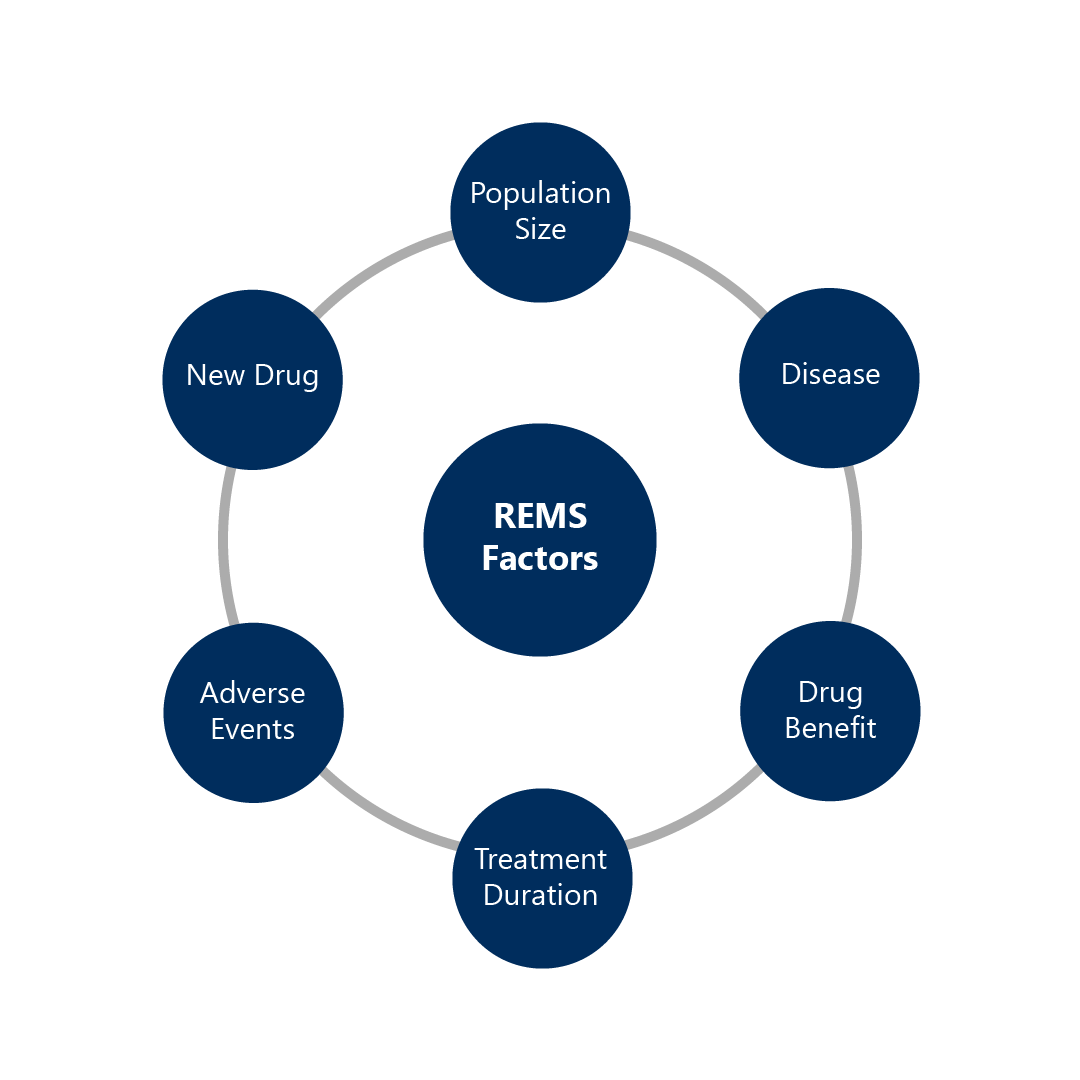
REMS Programs are decided on a case-by-case basis. The FDA considers six (6) factors to determine the need for a REMS Program.
All factors are considered together in the decision-making process rather than individually.
• Size of the population likely to use the drug
• Seriousness of the disease
• Expected benefit of the drug
• Expected duration of treatment
• Seriousness of known or potential adverse events
• Whether the drug is newly approved or an existing drug
REMS Compliance
FDA REMS Inspections are prioritized using a risk-based approach. Action will be taken if issues found during the inspection are not promptly and adequately addressed. Those actions can include product seizure, injunction, or financial penalties.
The FDA reviews surveillance reports to evaluate legal and regulatory compliance. Issues identified can result in regulatory action for noncompliance, which may include a warning letter to address serious safety concerns or mitigate risks to patients.
Making a good first impression is paramount for FDA REMS inspections and reviews.
FDA REMS Programs: Five Components for Compliance
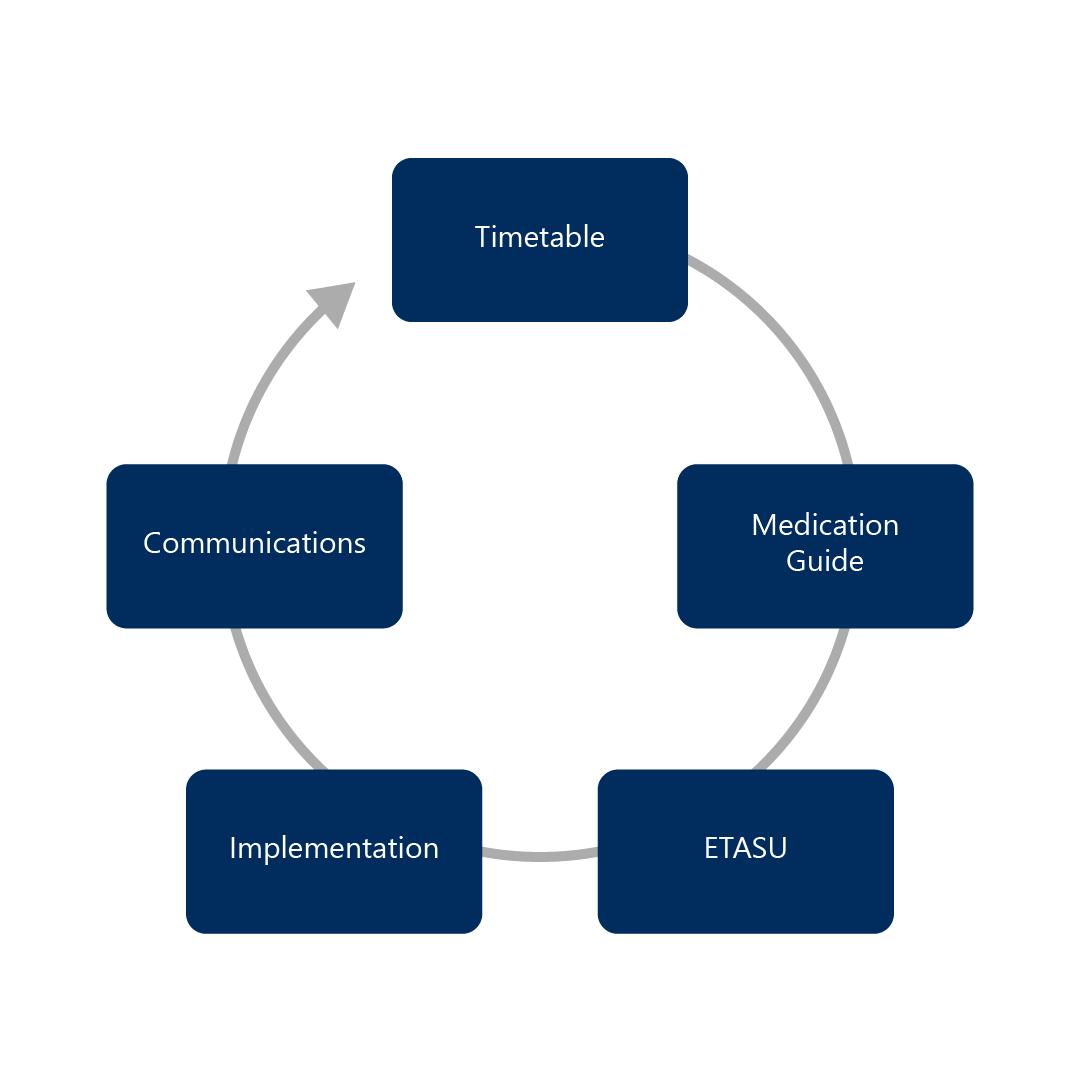
Five basic components must be included in a REMS Program.
Timetable for Submission of Assessments
The Timetable for Submission of Assessments states when Sponsors must assess how well the REMS is working and the FDA reporting requirements. Sponsors must include specific goals as evaluation metrics. These assessments compare the program results to the initial goals at 18 months, three years, and seven years.
Medication Guide
A medication Guide may be required to help avoid Serious Adverse Events (SAEs). The Medication Guide aims to inform patients about serious side effects before they decide to use a drug. Medication Guides are intended to be read and understood by the patient with adherence to prescribing and dosing, which is essential to the drug’s effectiveness measures.
Elements to Assure Safe Use (ETASU)
Many Elements to Assure Safe Use (ETASU) requirements focus on actions performed before the drug is dispensed or prescribed to a patient, such as certifications and dispense-only requirements. Other elements, such as patient monitoring, may be required for the duration of the treatment. The certification ensures that the healthcare providers know any serious risks and how to look for and control potential adverse events associated with the drug. REMS programs can require a pharmacist or anyone else who dispenses the drug to be certified (not only the prescribing physician).
Implementation System
An implementation System to track certain ETASU elements may be required. Sponsors are tasked to ensure reasonable steps to monitor and evaluate those in the healthcare system responsible for implementing necessary, safe use measures.
Communication Plan
A Communication Plan educates and raises awareness of the risks associated with the drug’s use. They are similar to Medication Guides, except they are directed to healthcare providers instead of patients. In addition to risk awareness education, Communication Plans alert healthcare providers of any changes in the REMS program so they can adequately care for their patients taking a specific medication. Communication Plans can be letters sent directly to healthcare providers and often include distributions of information materials to professional societies.
Considerations for Implementation of a REMS Program
The FDA can require a REMS program if they determine that safety measures are needed beyond drug labeling.
Sponsors are responsible for developing their REMS program for their specific drug or if it falls within a class of drugs requiring a REMS. Each REMS has specific safety measures unique to the risks associated; no two REMS programs are identical. There are commonalities for the successful implementation and management of a REMS program.
REMS are not designed to mitigate all adverse events of a medication. The focus is on preventing, monitoring, and/or managing a specific serious risk by educating prescribers, pharmacists, and patients to reduce the frequency or severity of the adverse event occurrence.
It can be challenging to implement REMS Programs, and they require specific components to remain in compliance and avoid FDA observations. The resources, time, and cost of a REMS Program must be evaluated prior to implementation.
Partnerships with REMS Program providers can alleviate some of these burdens from the pharmaceutical companies and place the workload on the organizations, with expertise, in delivering the successful components in compliance with the FDA REMS requirements.
The MMS Team can be your primary partner for managing the audits, CAPAs, and annual reporting requirements or completing annual surveillance reporting! Our experienced and knowledgeable team can help you implement these processes. Get in touch with us here.











